Our Science
Committed to changing the landscape of cancer and other serious diseases
About the Cell Cycle in Cancer
Loss of control of the cell cycle lies at the heart of cancer. In normal cells, a complex set of interacting proteins tightly regulates progression through the phases of the cell cycle. Specific isoforms of cyclin-dependent kinases (CDKs) and polo-like kinases (PLKs), are some of the key regulators of cell cycle checkpoints. If checkpoint control events are not completed correctly, cancer cells may commit suicide by a process of programmed cell death called apoptosis. We seek to enhance and facilitate apoptotic outcomes to treat patients with cancer.
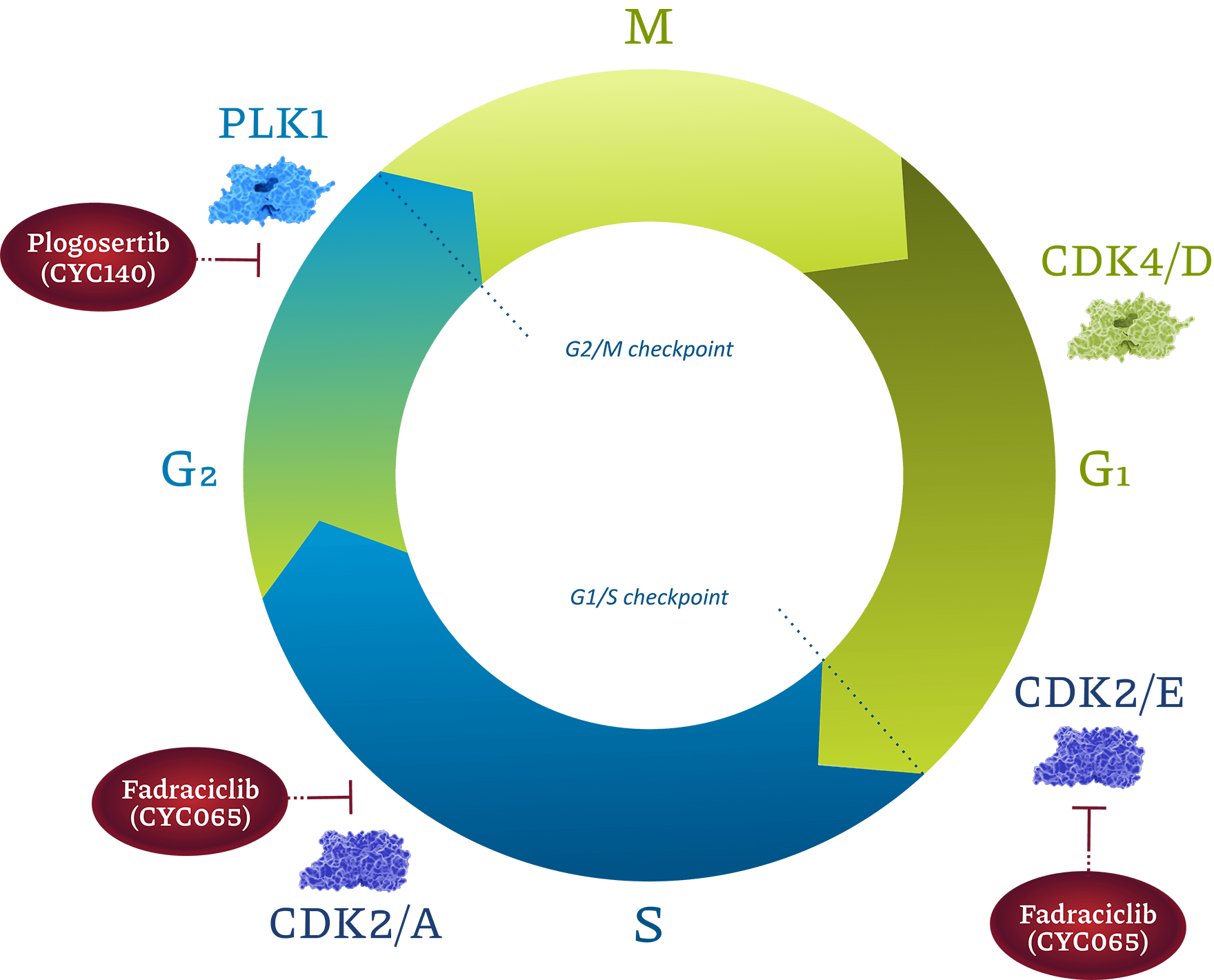
Transcriptional Regulation: Fadraciclib (formerly CYC065), a CDK2 and CDK9 inhibitor
A highly selective and potent amino purine inhibitor of CDK2 and CDK9, available both orally and intravenously.
Epigenetic/Mitosis Regulation: Plogosertib (formerly CYC140), a PLK1 inhibitor
A highly selective and potent inhibitor of PLK1, available both orally and intravenously.